Clinical trials today involve multiple sites, stakeholders, and systems, which makes operational coordination one of the biggest challenges in modern research. Notably, more than 500 global organisations already rely on Clinical Trial Management Systems (CTMS) to manage this complexity and bring structure to clinical trial operations.
Basically, a purpose-built CTMS provides a centralised system to plan, track, and manage clinical trial activities across their full lifecycle. It helps avoid fragmented workflows, scattered data, scheduling conflicts, and limited visibility across study operations, enabling research teams to maintain control, coordination, and compliance throughout the trial.
AQ’s CTMS guide provides updated insights on:
- CTMS definition, purpose, and role in clinical research operations
- How CTMS functions across study setup, participant tracking, scheduling, and execution workflows
- End-to-end CTMS modules supporting study planning, recruitment, visit management, financial tracking, and reporting
- Roles and teams that rely on CTMS across sponsors, CROs, sites, and coordinators
- Operational, financial, and coordination advantages of using a CTMS
- How CTMS enforces compliance through audit trails, role-based access, and regulatory controls
- Differences between CTMS and other clinical research systems including EDC, CDMS, QMS, eTMF, and Excel
- How CTMS integrates with clinical systems to maintain connected research operations
- Key factors to evaluate when selecting the right CTMS software
- What is AQ CTMS and how it offers a regulated research platform
What is a CTMS - Clinical Trial Management System?
A Clinical Trial Management System (CTMS) is a purpose-built software platform used to plan, coordinate, and oversee clinical trial operations across their full lifecycle. It brings study activities into one structured environment where teams maintain control over execution, visibility across sites, and alignment with regulatory requirements.
CTMS serves as the operational backbone of clinical research by connecting all core activities within a single system. Research teams rely on CTMS to manage:
- Study setup, timelines, and milestone planning
- Participant enrolment, tracking, and progression
- Visit scheduling aligned with protocol windows
- Site coordination, workload, and resource allocation
- Financial tracking including budgets, payments, and cost per visit
- Compliance controls such as audit trails, electronic records, and role-based access
- Reporting, dashboards, and real-time operational insights
This structured approach ensures that study activities remain connected, traceable, and consistently executed across teams and locations. Each action, update, and decision is recorded within the system, which strengthens accountability and supports inspection readiness throughout the trial.
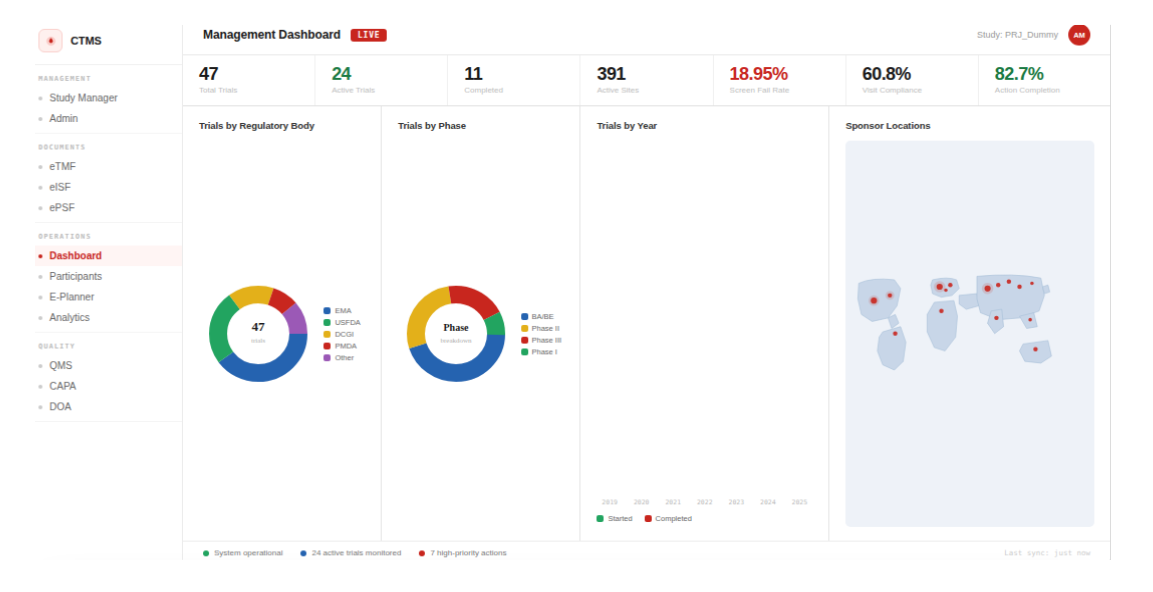
CTMS also enables continuous visibility into study performance. Research teams monitor recruitment flow, visit adherence, site activity, and operational signals in real time, which helps guide timely decisions and maintain progress across the study lifecycle.
In practice, CTMS creates a controlled operational environment where clinical trials move forward with clarity, coordination, and compliance at every stage.
Also Read: What Is Clinical Research Software and How to Choose the Right One?
How a CTMS Works in Clinical Trial Operations?
Let’s suppose a clinical research site starts a Phase II hypertension study with 80 participants. The study includes screening, baseline, and four follow-up visits, each defined with exact timelines and allowed visit windows.
Dr. Smith is assigned as the Principal Investigator, and Emma, the study coordinator, sets up the study inside the CTMS. She defines visit schedules with specific day ranges, assigns staff roles for each visit, and configures appointment slots based on room capacity and staff availability.
A participant, John Miller, completes screening and is enrolled into the study. The system immediately maps his full visit schedule based on the protocol.
- Emma books John’s baseline visit within the defined window after checking available slots in the planner
- The system schedules all upcoming visits with minimum and maximum allowed windows to maintain protocol compliance
- Automated email and SMS reminders are sent before each visit using predefined templates
- After each visit, John’s status updates automatically, and the next visit becomes active in the schedule
- Each visit records predefined payment and cost values, which are tracked against the study budget
- Staff roles assigned to the visit ensure that required personnel and time allocation remain structured
- A delay occurs before Visit 2; the system flags it as an out-of-window risk and alerts Emma
- Emma reschedules within the allowed window, and the system logs the activity with full traceability
- Dashboards update in real time, showing enrolment progress, visit adherence, staff utilisation, and site capacity
This scenario shows how CTMS connects study setup, participant management, scheduling, communication, financial tracking, and compliance within one controlled system. Each activity follows protocol-defined rules and remains visible across the research team.
CTMS enables clinical trials to operate with structured execution, coordinated workflows, and continuous oversight across every stage of the study.
Also Read: How Much Does a Clinical Trial Management System Cost?
End-to-End CTMS Modules Across the Clinical Trial Lifecycle
CTMS operates as a connected system of modules that manage study execution, participant activity, documentation, compliance, and oversight across the clinical trial lifecycle.
Core CTMS Operational Modules
- Study & User Management
Defines study structure, assigns investigators and coordinators, and controls roles, permissions, and study access. - Participant Management
Maintains participant records, enrolment status, clinical details, and study progression. - Study Scheduling & Visit Configuration
Structures protocol-driven visits with defined time points, window periods, and cohort logic. - E-Planner (Appointment & Visit Management)
Schedules and tracks participant visits using slot-based booking, calendars, and real-time availability. - Communication Management
Automates email and SMS communication for booking confirmations, reminders, and study updates. - Financial Management (Payments & Costs)
Tracks participant payments, visit compensation, and study-level cost allocation. - Capacity & Resource Management
Manages site locations, rooms, staff allocation, and occupancy for efficient scheduling. - Delegation of Authority (DOA) Management
Maintains a structured delegation log, mapping study tasks to authorized personnel with signatures, role validity, and full traceability for compliant study execution.
Document and Trial File Management Systems
- eISF (Electronic Investigator Site File)
Manages investigator site documents, regulatory files, and site-level records with version control and audit logs. - eTMF (Electronic Trial Master File)
Maintains central trial documentation across sponsors and CROs, ensuring completeness and inspection readiness. - ePSF (Electronic Pharmacy Site File)
Stores pharmacy-related documents, drug accountability records, and medication handling data.
Oversight, Quality, and Analytics Modules
- Quality Management System (QMS)
Manages quality processes, document approvals, change requests, and compliance workflows. - Analytics & Reporting
Provides dashboards on recruitment, visit adherence, participant status, staff utilisation, and study performance. - Audit & Activity Tracking
Records all system actions with time-stamped logs to ensure traceability and regulatory compliance.
A CTMS supports multiple stakeholders across clinical research by aligning responsibilities, improving visibility, and maintaining structured execution across studies.
Stakeholder | Role in Clinical Trials | How CTMS Supports Them |
Sponsors | Fund and oversee clinical trials, ensure timelines, budgets, and outcomes are met | Provides real-time visibility into study progress, financial tracking, and overall performance across sites |
Contract Research Organizations (CROs) | Manage trial operations on behalf of sponsors across multiple studies and locations | Enables coordination across sites, standardises workflows, tracks milestones, and ensures consistent execution |
Principal Investigators (PIs) | Lead clinical studies at sites and ensure protocol adherence | Offers structured view of participant progress, visit schedules, and study activities for controlled oversight |
Study Coordinators | Handle daily trial operations including scheduling, participant management, and documentation | Supports visit scheduling, participant tracking, communication, and workflow management in one system |
Clinical Research Associates (CRAs) | Monitor site performance, data accuracy, and compliance | Provides access to study data, visit tracking, audit logs, and site activity for monitoring and reporting |
Site Staff (Nurses, Technicians, Support Staff) | Conduct study visits, manage participants, and perform clinical procedures | Aligns schedules, assigns tasks, tracks visit requirements, and ensures coordination across staff roles |
Regulatory and Quality Teams | Ensure compliance with regulatory standards and manage documentation | Maintains audit trails, document control, and compliance workflows for inspection readiness |
Data and Operations Managers | Oversee study performance, resource allocation, and reporting | Delivers dashboards, analytics, and operational insights for informed decision-making |
What are the Advantages of Using a CTMS (Clinical Trial Management System)?
- Centralised control over all clinical trial activities
- Real-time visibility across participants, sites, and study progress
- Structured study execution aligned with protocol requirements
- Improved participant tracking and visit adherence
- Efficient scheduling with reduced conflicts and delays
- Automated communication for reminders and updates
- Better coordination across teams and locations
- Financial transparency with tracked payments and study costs
- Strong compliance support with audit trails and role-based access
- Reduced manual work through workflow automation
- Faster decision-making with live dashboards and reports
- Improved data accuracy and consistency across systems
- Enhanced resource utilisation for staff and site capacity
- Scalable operations across multiple studies and sites
- Inspection readiness with complete traceability of study activities
How to Choose the Right CTMS Software?
Selecting a CTMS requires evaluating how well the system supports real study execution, compliance, and long-term scalability across clinical research operations.
Key factors to consider include:
- Study workflow alignment
Ability to configure study setup, visit schedules, protocol windows, and participant flow based on real trial requirements - User roles and access control
Support for role-based permissions across investigators, coordinators, monitors, and sponsors - Participant and site management capabilities
Centralised tracking of enrolment, participant status, site activity, and workload distribution - Scheduling and visit management
Structured appointment planning with calendar views, slot management, cohort handling, and capacity control - Financial tracking and budgeting
Management of study budgets, participant payments, visit-based costs, and financial visibility - Compliance and audit readiness
Built-in audit trails, document control, electronic records management, and regulatory alignment - Document and file management integration
Support for eISF, eTMF, and document workflows with version control and access tracking - System integration capabilities
Ability to connect with EDC, QMS, eTMF, eISF, ePSF, and other clinical systems for a unified environment - Reporting and analytics
Real-time dashboards for recruitment, visit adherence, site performance, and operational insights - Automation and communication
Automated reminders, notifications, and workflow triggers to reduce manual coordination - Scalability across studies and sites
Capacity to handle multi-site, multi-study environments with increasing data volume - Usability and adoption
Intuitive interface, structured navigation, and ease of use for different user roles - Data security and access management
Secure authentication, controlled access, and protection of sensitive participant data - Implementation and onboarding support
Ease of setup, training, configuration, and transition into ongoing clinical trial workflows - Vendor reliability and support
Ongoing technical support, updates, system reliability, and long-term product stability
AQ Trials: Unified CTMS Platform for Regulated Research Environments
AQ Trials is an all-encompassing CTMS platform that brings clinical trial execution, documentation, compliance, and oversight into a single connected system. It establishes a unified operational environment where study activities, participant management, scheduling, financial tracking, and regulatory processes operate in alignment rather than across disconnected tools.
It offers access to the following clinical research software in an end-to-end platform:
- Study Manager
- Participant Manager
- E-Planner
- Analytics
- eTMF
- eISF
- ePSF
- QMS
- CAPA Management
- DOA
How does AQ CTMS bring study execution, documentation, and oversight into one connected system?
AQ CTMS connects study execution, document management, and oversight within a single operational environment, where each activity remains linked to the study structure, participant lifecycle, and regulatory requirements. Study workflows, participant actions, documents, and quality processes operate within the same system, ensuring that execution and compliance progress together without fragmentation.
This connection is achieved through:
- Unified study configuration
Study setup, schedules, visit structures, and user roles are defined within one system and applied consistently across all modules - Linked participant and visit workflows
Participant enrolment, visit scheduling, and activity tracking remain connected to study protocols and timelines - Integrated document management (eISF/eTMF)
Study documents, investigator files, and regulatory records are directly associated with study activities and user roles - Embedded quality and compliance processes (QMS)
Deviations, change requests, approvals, and quality workflows are linked to operational events and study actions - Centralised audit trails and activity logs
Every action across execution, documentation, and oversight is recorded with full traceability - Real-time operational and compliance visibility
Dashboards provide a unified view of study progress, participant activity, document status, and quality metrics
This structure ensures that clinical trials operate within a connected system where execution, documentation, and oversight remain aligned throughout the study lifecycle.
In what way does AQ CTMS improve visibility across participants, sites, and ongoing study activity?
AQ CTMS improves visibility by centralising all study activities into a single system where participant progress, site operations, and study performance can be monitored in real time. Each action, from enrolment to visit completion, is recorded and reflected across dashboards, which allows research teams to maintain a clear and continuous view of what is happening across the study.
This visibility is achieved through:
- Real-time participant tracking
Displays enrolment status, visit history, upcoming appointments, attendance, and overall progression for each participant - Site-level performance monitoring
Tracks recruitment rates, visit adherence, staff workload, and capacity utilisation across sites - Centralised study dashboards
Provides live insights into study progress, milestones, and operational metrics within one interface - Visit and scheduling transparency
Shows appointment status, upcoming visits, delays, and out-of-window risks across participants - Cohort and group-level visibility
Organises participants into study groups and tracks their progress collectively - Staff and resource utilisation insights
Monitors how staff time, roles, and site resources are allocated and used - Deviation and activity alerts
Highlights missed visits, scheduling conflicts, and protocol deviations for immediate attention
This level of visibility enables research teams to identify issues early, adjust study activities in time, and maintain consistent progress across participants, sites, and the overall trial.
How does AQ CTMS support compliance, audit readiness, and regulatory traceability in clinical research?
AQ CTMS embeds compliance within everyday study operations by linking protocol execution, documentation, and user actions to controlled, traceable processes. Each activity across study setup, participant management, scheduling, and documentation is recorded with full context, which supports continuous audit readiness and regulatory alignment.
This support is achieved through:
- Comprehensive audit trails
Records every action with user identity, timestamp, and activity details across studies, participants, schedules, and documents - Role-based access control
Restricts system access based on defined user roles, ensuring that only authorised personnel can view or modify data - Protocol-aligned execution controls
Enforces visit schedules, window periods, and study workflows based on predefined protocol rules - Deviation tracking and reporting
Captures out-of-window visits, missed activities, and protocol deviations with structured reporting and visibility - Integrated document compliance (eISF/eTMF)
Maintains version-controlled documents, regulatory files, and investigator records linked to study activities - Quality process integration (QMS)
Manages change control, document approvals, and quality workflows connected to operational events - Mandatory document review and training controls
Ensures users complete required document reading or training before proceeding with study-related tasks - Secure authentication and controlled access
Uses structured login processes and access controls to protect sensitive study and participant data - Inspection-ready reporting and data access
Provides organised, exportable records of study activities, documents, and compliance logs for audits
This structured approach ensures that clinical trials maintain traceability across all actions, accountability across users, and readiness for regulatory inspections at any stage of the study lifecycle.
What makes AQ Clinical Research Software different from traditional CTMS tools?
AQ Clinical Research Software differs from traditional CTMS tools by operating as a connected execution system rather than a standalone tracking solution. It brings study workflows, participant activity, documentation, and quality processes into one environment, where each function is linked to the others and driven by structured configurations.
This difference is reflected in how the system is designed and used:
- Execution-driven system vs tracking-focused tools
Supports real study workflows such as scheduling, visit windows, staff allocation, and participant progression instead of only recording activities - Connected modules within one platform
Combines CTMS, document management (eISF/eTMF), and quality processes (QMS) into a single system rather than separate tools - Protocol-based configuration
Defines study schedules, visit rules, payments, and communication flows directly within the system to guide execution - Integrated scheduling and capacity management
Aligns participant visits with site availability, room capacity, and staff workload in real time - Built-in communication workflows
Automates participant and site communication through predefined templates and triggers - Linked financial tracking
Connects visit execution with payment and cost tracking at participant and study levels - Continuous compliance alignment
Embeds audit trails, document controls, and quality processes into operational workflows - Real-time operational visibility
Provides dashboards across participants, sites, staff, and study performance within one interface
Traditional CTMS tools primarily record and monitor study activities. AQ operates as a system where those activities are planned, executed, tracked, and governed within a unified structure, enabling more controlled and connected clinical trial operations.
How can AQ be introduced into existing clinical trial workflows without disrupting ongoing studies?
AQ CTMS can be introduced alongside existing processes through a phased approach that aligns with current study operations. The system allows teams to configure studies, users, and workflows in parallel, which enables gradual adoption while ongoing trials continue without interruption.
This transition is managed through:
- Parallel setup with existing workflows
Studies, schedules, users, and roles are configured in AQ while current systems remain active - Phased module adoption
Teams begin with core functions such as participant tracking and scheduling, then expand into documents, compliance, and analytics - Selective data migration
Active participant data, study details, and schedules are transferred in stages to maintain continuity - Role-based onboarding and training
Users are introduced to the system based on their responsibilities, ensuring smooth adoption across coordinators, investigators, and staff - Controlled rollout across sites or studies
Implementation can begin with selected studies or locations before expanding across the organisation - Workflow alignment with study protocols
Existing processes are mapped into system configurations to ensure consistency with current study execution - Minimal disruption to ongoing visits and schedules
Active appointments and participant timelines continue while new activities are managed within the system - Continuous monitoring during transition
Dashboards and reports help teams track adoption, identify gaps, and adjust workflows in real time
This approach ensures that AQ integrates into clinical research environments without interrupting ongoing studies, while gradually establishing a structured and connected operational system.
CTMS and Compliance
Compliance in clinical research operates as a continuous, structured process that spans protocol execution, documentation, data handling, and audit readiness. Notably, a Clinical Trial Management System (CTMS) establishes this structure by embedding regulatory controls, traceability, and governance directly into daily study operations.
CTMS aligns clinical trial activities with globally recognised regulatory frameworks and standards, including:
- FDA regulations for clinical investigations and electronic records
- EMA guidelines for trial conduct and oversight within the EU
- ICH-GCP standards for ethical and scientific quality in clinical trials
- 21 CFR Part 11 for secure electronic records and signatures
- GDPR for data privacy and protection across participant information
Structured Compliance Through CTMS
CTMS embeds compliance into core workflows rather than treating it as a separate activity. Each action within the system follows defined rules and remains fully traceable.
- Role-based access control. User permissions are assigned based on roles such as investigator, coordinator, monitor, or administrator. Access to study data, documents, and actions remains restricted and controlled.
- Audit trails and activity logs. Every system action is recorded with user identity, timestamp, and activity details. Study creation, updates, scheduling changes, and participant actions remain fully traceable.
- Protocol-driven execution. Visit schedules, window periods, and study workflows are configured according to protocol requirements. Each participant progression follows predefined rules, ensuring adherence throughout the study.
- Deviation tracking and monitoring. Out-of-window visits, missed appointments, and protocol deviations are captured and reported through system analytics for timely review and corrective action.
Document and Record Compliance
CTMS works closely with document management systems to maintain structured and compliant records across the trial.
- eISF (Electronic Investigator Site File)
Stores site-level regulatory documents, investigator records, and study files with version control and access tracking - eTMF (Electronic Trial Master File)
Maintains central trial documentation across sponsors and CROs, ensuring completeness and inspection readiness - QMS (Quality Management System)
Manages document approvals, change requests, CAPA processes, and quality workflows - Document compliance enforcement
Systems can require mandatory reading, training completion, or document acknowledgment before allowing users to proceed with study tasks
Data Integrity and Security
CTMS ensures that study data remains accurate, consistent, and protected throughout its lifecycle.
- Controlled data entry and validation rules
- Secure authentication mechanisms and access codes
- Encrypted storage and controlled data sharing
- Version-controlled updates with full history tracking
All these controls support data integrity principles required for regulatory acceptance and inspection readiness.
Inspection Readiness and Oversight
CTMS prepares research teams for audits and inspections by maintaining a complete, organised, and accessible record of all study activities.
- Centralised access to study data, documents, and logs
- Real-time dashboards for monitoring compliance status
- Exportable reports for audits and regulatory submissions
- Transparent tracking of user actions and study changes
Compliance as a Core Operational Function
CTMS transforms compliance from a reactive task into an integrated operational layer. Study execution, documentation, communication, and reporting all operate within controlled boundaries defined by regulatory standards.
This approach ensures that clinical trials maintain:
- consistency in execution
- traceability across all actions
- accountability across teams
- readiness for audits at any stage
So it is clear that CTMS enables research organisations to manage clinical trials with structured governance, controlled workflows, and continuous compliance aligned with global regulatory expectations.
And if you would like to explore further how AQ can revolutionise your clinical trials—reach out to us for a free online consultation.
How Does CTMS Interact With Other Systems in Clinical Trials?
Clinical trials operate through multiple specialised systems, each managing a specific function. A CTMS connects these systems to ensure that study execution, data flow, documentation, and compliance remain aligned across the trial lifecycle.
CTMS acts as the operational hub that links activities across systems while maintaining a consistent view of study progress, participant activity, and site performance.
Key System Interactions
- With Electronic Data Capture (EDC)
CTMS aligns visit schedules, participant enrolment, and site activity with data collected during study visits. EDC captures clinical data, while CTMS tracks when and where that data is generated. - With Clinical Data Management System (CDMS)
CTMS provides operational context such as visit timelines and participant status, while CDMS validates, cleans, and prepares collected data for analysis. - With Electronic Trial Master File (eTMF)
CTMS links study activities with required documents. eTMF stores regulatory files, approvals, and trial documentation, ensuring completeness and inspection readiness. - With Quality Management System (QMS)
CTMS tracks operational events, while QMS manages deviations, change control, CAPA processes, and quality workflows linked to those events. - With eISF (Electronic Investigator Site File)
CTMS connects participant visits and site activity with investigator site documents, training records, and compliance files. - With Financial and Payment Systems
CTMS tracks study budgets, visit-based payments, and cost allocation, while external financial systems handle processing and accounting. - With Laboratory Information Management Systems (LIMS)
LIMS is used to manage and track laboratory samples and test results. CTMS integration with LIMS allows for real-time tracking of sample collection and results. Well, this significantly helps in keeping a quick check on the progress of the trial while ensuring data accuracy at every step.
What This Integration Enables?
- Unified visibility across operations, data, and documentation
- Consistent participant and visit tracking across systems
- Improved data accuracy through aligned workflows
- Faster issue identification and resolution
- Stronger compliance with complete traceability
Operational Perspective
CTMS does not replace other systems. It connects them. Each system continues to perform its specialised function, while CTMS ensures that all activities remain coordinated, visible, and aligned within a single operational framework.
This interconnected approach enables clinical trials to run with structured execution, controlled data flow, and integrated oversight across every stage.
CTMS VS Other Clinical Trial Research Software
It is important to understand the difference between a unified CTMS system and other tools that clinical researchers use across the trial lifecycle, as each system serves a distinct purpose within study execution, data handling, compliance, and documentation.
Aspect | CTMS (Clinical Trial Management System) | Electronic Data Capture (EDC) |
Primary Purpose | Manages clinical trial operations and study execution | Captures and stores clinical trial data from study visits |
Focus Area | Operational management (sites, participants, visits, workflows) | Data collection and clinical data entry |
Core Function | Plans, tracks, and coordinates study activities | Records patient data in electronic case report forms (eCRFs) |
Data Type | Operational data (scheduling, enrolment, payments, site activity) | Clinical data (lab results, observations, outcomes) |
Users | Study coordinators, investigators, sponsors, CROs | Site staff, data entry personnel, clinical data managers |
Role in Workflow | Drives study execution and coordination across sites | Collects and structures data generated during study visits |
Compliance Role | Tracks protocol adherence, audit trails, and operational oversight | Ensures accurate, validated, and structured clinical data capture |
Integration | Integrates with EDC, eTMF, QMS, and other systems | Integrates with CTMS and CDMS for data flow and validation |
Example Use | Scheduling participant visits and tracking study progress | Entering patient vitals, lab data, and clinical observations |
Outcome | Provides visibility and control over trial operations | Provides clean, structured datasets for analysis and reporting |
CTMS VS CDMS
Aspect | CTMS (Clinical Trial Management System) | Clinical Data Management System (CDMS) |
Primary Purpose | Manages clinical trial operations and execution | Manages, cleans, and validates collected clinical data |
Focus Area | Operational workflows and study coordination | Data processing, validation, and quality control |
Core Function | Tracks study progress, participants, visits, and site activity | Ensures data accuracy through validation checks, queries, and data cleaning |
Data Type | Operational data (enrolment, scheduling, payments, site performance) | Clinical data (patient records, lab results, trial outcomes) |
Users | Study coordinators, investigators, sponsors, CROs | Data managers, biostatisticians, clinical data teams |
Role in Workflow | Drives execution and coordination of the trial | Prepares collected data for analysis and regulatory submission |
Compliance Role | Maintains audit trails, protocol adherence, and operational oversight | Ensures data integrity, consistency, and validation against protocols |
Integration | Connects with EDC, CDMS, eTMF, and QMS | Receives data from EDC and integrates with CTMS for context |
Example Use | Monitoring recruitment progress and scheduling visits | Running data validation checks and resolving data discrepancies |
Outcome | Provides visibility and control over trial operations | Produces clean, reliable datasets for statistical analysis |
CTMS VS QMS
Aspect | CTMS (Clinical Trial Management System) | Quality Management System (QMS) |
Primary Purpose | Manages clinical trial operations and execution | Manages quality processes, compliance, and governance |
Focus Area | Study workflows, participants, visits, and site coordination | Quality control, document approvals, audits, and CAPA processes |
Core Function | Tracks and coordinates day-to-day study activities | Ensures processes follow defined quality standards and regulatory requirements |
Data Type | Operational data (scheduling, enrolment, payments, site activity) | Quality and compliance data (deviations, change requests, audits, CAPA) |
Users | Study coordinators, investigators, sponsors, CROs | Quality teams, compliance officers, auditors, management |
Role in Workflow | Drives execution and operational control of the trial | Monitors and enforces quality across processes and documentation |
Compliance Role | Maintains audit trails, protocol adherence, and operational visibility | Manages audits, document control, approvals, and corrective actions |
Integration | Connects with QMS, EDC, eTMF, and other systems | Integrates with CTMS to align quality processes with study activities |
Example Use | Scheduling visits and tracking participant progress | Managing deviations, approving documents, handling change control |
Outcome | Ensures studies run efficiently and according to plan | Ensures studies meet quality standards and regulatory expectations |
Aspect | CTMS (Clinical Trial Management System) | Electronic Trial Master File (eTMF) |
Primary Purpose | Manages clinical trial operations and execution | Manages and stores essential trial documents and records |
Focus Area | Study workflows, participants, visits, and site coordination | Document collection, organisation, and regulatory documentation |
Core Function | Tracks and coordinates study activities across sites and teams | Maintains complete, version-controlled trial documentation for compliance |
Data Type | Operational data (enrolment, scheduling, payments, site activity) | Regulatory documents (protocols, consent forms, approvals, reports) |
Users | Study coordinators, investigators, sponsors, CROs | Regulatory teams, document specialists, sponsors, auditors |
Role in Workflow | Drives execution and operational visibility of the trial | Ensures all required documents are complete, organised, and inspection-ready |
Compliance Role | Maintains audit trails, protocol adherence, and activity tracking | Ensures document completeness, version control, and regulatory compliance |
Integration | Connects with eTMF, EDC, QMS, and other systems | Integrates with CTMS to link documents with study activities |
Example Use | Scheduling visits and tracking study progress | Storing investigator brochures, ethics approvals, and study documents |
Outcome | Provides control and visibility over trial execution | Provides a complete, auditable record of trial documentation |
CTMS VS Excel
Aspect | CTMS (Clinical Trial Management System) | Microsoft Excel |
Purpose | Executes and manages clinical trial operations | Tracks data manually in spreadsheets |
Approach | System-driven workflows with defined processes | User-driven tracking without built-in structure |
Data Handling | Centralised, connected, and continuously updated | Static, file-based, and manually updated |
Study Execution | Supports scheduling, participant tracking, and site coordination | Requires manual tracking across multiple sheets |
Visibility | Real-time dashboards across studies, sites, and participants | Limited visibility confined to individual files |
Collaboration | Multi-user access with role-based permissions | File sharing with version conflicts and duplication risks |
Compliance | Built-in audit trails, access control, and traceability | No native compliance controls or audit tracking |
Error Control | Validation rules and automation reduce operational errors | High risk of manual errors and inconsistencies |
Scalability | Handles multi-site, multi-study environments efficiently | Becomes difficult to manage as complexity increases |
Integration | Connects with EDC, QMS, eTMF, and other systems | Operates as a standalone tool with limited integration |
Outcome | Structured, controlled, and scalable trial execution | Basic tracking with limited control and oversight |
Explore AQ CTMS Now
Request a demo to see how AQ supports structured clinical trial execution, integrated workflows, and real-time operational oversight across your research environment.
We will walk you through how AQ brings CTMS, eTMF, eISF, ePSF, QMS, and CAPA management into one connected platform—covering study setup, participant management, visit scheduling, document control, quality processes, and audit-ready traceability in real clinical trial operation
Frequently Asked Questions
CTMS software means Clinical Trial Management System in medical terms. It’s a specialised software designed to collectively manage and accelerate the planning, tracking, and execution of clinical trials in the field of medical research.
The clinical researchers use the CTMS to effectively streamline and optimise the operational aspects of clinical trials. This is because a clinical trial management system makes it hassle-free and efficient to manage tasks such as participant recruitment, scheduling, data collection, budgeting, and reporting. All while ensuring compliance.
When you choose a CTMS, look for features like real-time protocol management, site management, regulatory compliance, integration capabilities, user-friendliness, scalability, and cost-effectiveness. All this is necessary to ensure quick and reliable trial processes.
EDC (Electronic Data Capture) focuses on collecting and managing clinical trial data, often related to patient records and outcomes. Whereas CTMS (Clinical Trial Management System) oversees the overall planning, coordination, and tracking of the entire clinical trial, managing operational aspects beyond data capture. The EDC and CTMS integration allows for a seamless flow of information between the systems, improving efficiency and data accuracy.
CTMS (Clinical Trial Management System) is primarily focused on project management aspects of clinical trials, including startup, documents, payments, and monitoring. EDC (Electronic Data Capture) focuses on collecting patient data.
Both CTMS and EDC are often used together in clinical studies. CTMS manages project-related aspects, while EDC collects patient data. Integration of a subset of EDC data into CTMS is beneficial for high-level screening, enrollment tracking, subject visit progress, data collection status, and end-of-study disposition.
CTMS tracks anonymized subject records, allowing the study management team to monitor enrollment at a high level. This data is valuable for site payment tracking, visit report authoring, and other project management activities.
Worksheets are quick and easy but have limitations as the study grows. CTMS offers advantages such as collaboration with user roles, integrated and consistent data, secure and reliable access, and compliance with regulatory requirements.
CTMS provides a secure environment for team collaboration with controlled user access. Users can be restricted to specific data views and studies, ensuring a collaborative yet controlled working environment.
The CTMS acts as a warehouse of data, applying consistency across data views within a study and across different studies. This ensures that data is integrated and applied consistently throughout the organization.
CTMS provides controlled user accounts, restricting data access to authorized users only. High availability cloud computing technologies, backups, and redundancy ensure data availability when needed.
CTMS enforces controls for 21 CFR Part 11 compliance seamlessly in the background. These controls include an audit trail, electronic signatures, data archival, and user account controls, ensuring the system’s compliance with regulatory requirements.
Our pricing strategy is based on the size of the organisation and the ability to select the applications which are required. We are very competitively priced and our growth strategy is for every site to be able to afford a CTMS solution.
Related Articles
Industry Discussions
What is the Difference Between CTMS and eTMF
In clinical trial research management, the Clinical Trial Management System (CTMS) and the Electronic Trial Master File (eTMF) play distinct yet important roles. CTMS focuses on operational efficiency—overseeing site management and subject recruitment. In contrast, eTMF is an electronic repository—emphasizing document integrity, version control, and audit trails. All in real-time.
AQ Platform Updates
CTMS VS EDC - What’s their Role and Difference in Clinical Research
Clinical trial management systems (CTMS) and electronic data capture (EDC) software are designed to simplify and automate the intricate processes of clinical research. CTMS focuses on the management and coordination of the trial’s administrative aspect — whereas EDC software is dedicated to the collection and management of patient data. Both serve different but complementary roles, ensuring efficiency, accuracy, and success.